Summary: While current weight loss blockbusters like GLP-1s focus on suppressing appetite, researchers have uncovered a completely different strategy: increasing energy expenditure by “building out” the body’s natural heat-generating tissues.
The study revealed how a protein called SLIT3 acts as a “split signal” for the growth of nerve tissue and blood vessels important in brown fat. Without this infrastructure, brown fat cannot receive the brain’s “heat” signal or the nutrients it needs to burn calories. These findings suggest that obesity can be treated by increasing the body’s internal “metabolic sink” rather than simply eating less.
Key facts
- “Split Signal”: When brown fat cells secrete SLIT3, the enzyme is called MP1 split into two fragments. A fragment of the growth blood vessels (provide fuel), while others extend the tea nerve (providing an “on” switch).
- Metabolic sink: Activated brown fat acts as a “sink,” drawing in glucose and lipids from the bloodstream to produce heat (thermogenesis) instead of storing them as white fat.
- PLXNA1 receptor: The researcher identified PLXNA1 as a special docking station for SLIT3 that regulates nerve density. Mice lacking these receptors are unable to maintain their body temperature in cold temperatures because their brown fat lacks the “wires” to hear brain signals.
- Human evidence: Analyzing fat samples from more than 1,500 people, the team found that SLIT3 gene expression is closely linked to metabolic health, inflammation, and insulin sensitivity in individuals with obesity.
Source: turtle
Researchers have determined how a key protein activates brown fat by dilating blood vessels and nerves in heat-generating tissue.
Findings, published in Nature Communicationpoints to a potential strategy to treat obesity that deviates from the current approach of suppressing appetite.
Most of the fat in our body is white fat, which stores excess energy and, at too high a level, can lead to obesity. Humans and other mammals also have small amounts of brown fat, a specialized tissue that regulates body temperature and is closely linked to weight loss and metabolic health. When activated by cold exposure, brown fat uses the body’s resources such as glucose and lipids to generate heat, a process called thermogenesis.
“During thermogenesis, all chemical energy is dissipated as heat instead of being stored in the body as white fat,” said Farnaz Shamsi, assistant professor of molecular pathobiology at NYU College of Dentistry and senior author of our study.
“By quickly taking and using fuel sources from our body and the food we eat, brown fat acts like a metabolic sink that draws nutrients and prevents them from being stored.”
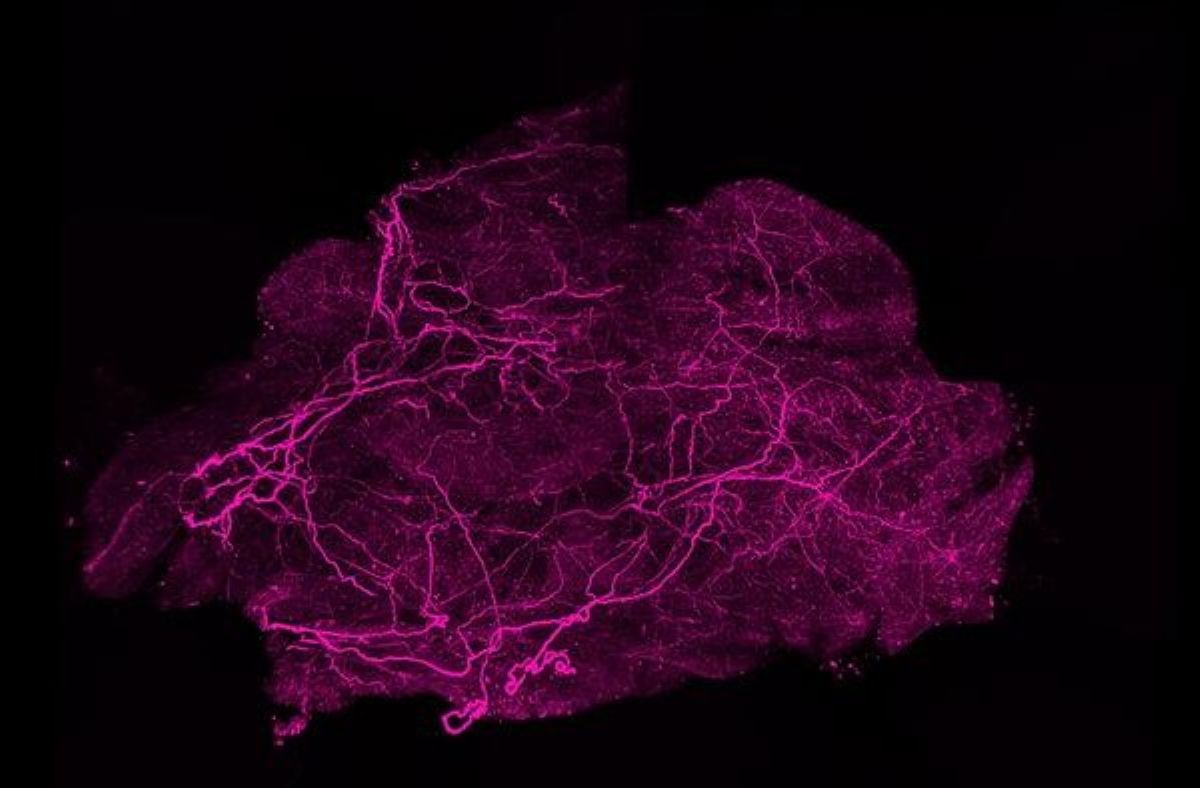
Brown fat has a complex and dense network of nerves and blood vessels that are essential for its function. Nerves allow brown fat to communicate with the brain; when the brain feels cold, it quickly signals to activate brown fat.
Blood vessels supply brown fat with oxygen and nutrients to generate heat, and then distribute this heat throughout the body. While research on brown fat has focused on stimulating fat cells to produce heat, less is known about how this basic tissue functions.
Shamsi’s lab previously used single-cell RNA sequencing to identify SLIT3, a protein secreted by brown fat cells, which they thought might play a role in how fat cells communicate. When generated, SLIT3 will be split into two different fragments.
ignoble Nature Communication The study, using a combination of approaches in human and mouse cells, researchers found the enzyme, BMP1, which is responsible for cleaving SLIT3 into two. It was also determined that two SLIT3 fragments control different processes: one grows blood vessel tissue, while the other expands nerve tissue.
“It works as a split signal, which is an elegant evolutionary design in which two components of a single factor independently regulate different processes that must be tightly coordinated in space and time,” noted Shamsi.
Additionally, the researchers identified a receptor, PLXNA1, that binds to one of the SLIT3 fragments to control brown fat nerve tissue. In studies in mice – which normally have very active brown fat and can tolerate cold temperatures for long periods – removing SLIT3 or the PLXNA1 receptor from brown fat causes the mice to become sensitive to cold and have difficulty maintaining their body temperature. A closer look at brown fat tissue that is missing SLIT3 or its receptor reveals that it lacks the proper neural structure and blood vessel density.
To see if the findings translate to humans, the researchers examined fat tissue samples from more than 1,5000 people, some of whom suffered from obesity. Focusing on the gene that produces SLIT3, which previous studies have shown to be associated with obesity and insulin resistance, they found that SLIT3 gene expression can regulate fat tissue health, inflammation, and insulin sensitivity in people with obesity.
“It really got our attention, because it suggests that this pathway may be relevant in human obesity and metabolic health,” Shamsi said.
While most weight loss drugs—including GLP-1s—suppress appetite, decreasing the amount of food people eat and therefore the amount of energy stored, treatment involving brown fat has the potential to increase energy expenditure.
This new understanding of what happens inside brown fat—including how SLIT3 binds to and binds to receptors to control nerves and blood vessels—highlights several processes that could potentially be exploited for its therapeutic potential.
“Our research shows that just having brown fat is not enough – you need the right infrastructure in the network for heat production,” says Shamsi.
Additional study authors include Tamires Duarte Afonso Serdan, Heidi Cervantes, Benjamin Frank, Akhil Gargey Iragavarapu, Qiyu Tian, Daniel Hope, and Halil Aydin of NYU College of Dentistry; Chan Hee Choi and Paul Cohen from Rockefeller University; Anne Hoffmann and Matthias Blüher from the University of Leipzig; Adhideb Ghosh and Christian Wolfrum from ETH Zurich; Matthew Greenblatt of Weill Cornell Medical College; and Gary Schwartz of the Albert Einstein College of Medicine.
Funding: The research was supported in part by the National Institutes of Health (K01DK125608, R03DK135786, R01DK136724, RC2DK129961, R35GM150942), the G. Harold and Leila Y. Mathers Charitable Foundation, The American Heart Association (274CDA) (2018). Diabetes Center, NYU Department of Dentistry of Molecular Pathobiology, and the Boettcher Foundation.
Key Questions Answered:
A: As senior author Farnaz Shamsi pointed out, “just having brown fat is not enough.” If your brown fat doesn’t have the right “infrastructure” — meaning enough nerves to hear brain signals and enough blood vessels to receive oxygen — it remains inactive. It’s like having a high-performance engine without a fuel line or ignition switch.
A: Most of the current drugs (GLP-1s) work by telling your brain that you are not hungry, which reduces it incoming energy. This SLIT3 line is about improving energy out. By “increasing” your brown fat, you are basically changing up your body’s internal thermostat to burn through stored white fat and blood sugar.
A: We may not have to grow More brown fat, but quite optimize what we have. By exploiting the SLIT3-PLXNA1 pathway, scientists hope to develop therapies that “renovate” existing brown fat, making it more efficient to absorb and burn excess nutrients.
Editor’s Note:
- This article was edited by the editors of Neuroscience News.
- Journal papers are reviewed in full.
- Additional context was added by our staff.
About this neurology and aging research news
author: Rachel Harrison
Source: turtle
Contact: Rachel Harrison – NYU
Image: Image credited to Shamsi Lab, NYU College of Dentistry
Original Research: Open access.
“SLIT3 fragments orchestrate neurovascular expansion and thermogenesis in brown adipose tissue” by Tamires Duarte Afonso Serdan, Heidi Cervantes, Benjamin Frank, Akhil Gargey Iragavarapu, Qiyu Tian, Daniel Hope, Chan Hee J. Choi, Anne Hoffmann, Adhideb Ghosh, Christian Wolfrum, Matthew B. Greenherblathia, Paul Co. Schwartz & Farnaz Shamsi. Nature Communication
DOI: 10.1038/s41467-026-70310-9
abstract
SLIT3 fragments orchestrate neurovascular expansion and thermogenesis in brown adipose tissue
Brown adipose tissue is an evolutionary innovation in placental mammals that regulates body temperature through adaptive thermogenesis. Cold exposure activates brown adipose tissue thermogenesis through the coordinated induction of brown adipogenesis, angiogenesis, and sympathetic innervation; however, how this process is combined remains unclear.
Here, we show that the Slit guidance ligand 3 (SLIT3) fragment drives crosstalk among adipocyte progenitors, endothelial cells, and sympathetic nerves. Adipocyte progenitors secrete SLIT3, which is split into functionally different SLIT3-N and SLIT3-C fragments that independently promote angiogenesis and sympathetic innervation.
We identified PLXNA1 as a receptor for SLIT3-C and demonstrated its important role in sympathetic innervation of brown adipose tissue. Moreover, we identified BMP1 as the first SLIT protease described in a vertebrate.
Coordinated neurovascular expansion mediated by different SLIT3 fragments provides a bifurcated yet integrated mechanism that ensures a synchronized brown adipose tissue response to environmental challenges.
Finally, this study reveals a previously unrecognized role for adipocyte progenitors in regulating tissue innervation.
#Split #Protein #Builds #Infrastructure #Weight #Loss #Neuroscience #News