Scientists are still discovering how weight loss “wonder drugs” like Ozempic and Zepbound reshape the body.
Researchers at the Salk Institute have found that long-term use of this drug reprograms cells and produces long-lasting and potentially beneficial changes that extend far beyond weight loss.
Sam Van de Velde, PhD, , first author of a paper on the subject, reviewed in the Proceedings of the National Academy of Sciences, has spent five years looking for “the next piece of the puzzle.”
“They seem to be doing a lot of things at once. Scientists don’t really like the term ‘wonder, miracle’. We prefer not to wonder about anything – just understand everything,” Van de Velde said.

More than 30 million Americans use this drug, according to KFF. Gallup reports its use will double from 2024 to 2025, and is still rising.
Originally developed to treat type 2 diabetes, this drug mimics a chemical signal created by the gut after eating, known as GLP-1. Today, they are better known for the side effects of weight loss, which restricts appetite, leading to changes in eating habits.
Stability is a key difference between natural and injected chemicals, meaning the drug lasts longer in the body. Scientists first found a stable version in the saliva of the Gila monster, one of only two known venomous lizard species in the world.
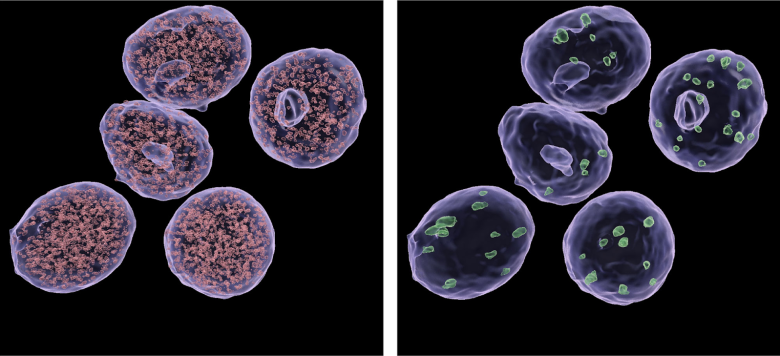
When this chemical reaches the pancreas, it is picked up by a small cluster of beta cells. This releases insulin, which helps cells absorb sugar to regulate blood sugar; Think of them as sugar sensors.
“Type 2 diabetes occurs when your blood sugar and the level of fat particles in your blood increases; this is toxic to beta cells, they decrease in health. Stable GLP-1 drug improves the health of beta cells and increases their insulin production capacity,” said Van de Velde.
The drug must be attached to the beta cell for this communication to occur. This attachment leads to a process called phosphorylation – a small chemical modification that adds a phosphate group to the protein, affecting how it works.
The team focused on a specific protein complex, Med14, that mediates gene activation. In this case, it is part of the body’s insulin production in the pancreas. It is known as “conserved protein”.
“There’s Med14 in chickens, yeast, all kinds of fungi, and, of course, people. They can’t survive without it; the body can’t develop – that’s how important it is,” says Van de Velde.
“We found that in this protein, a small chemical modification occurs when the cells are treated with the drug GLP-1, which allows hundreds of genes to be simultaneously activated. We call this the genomic response, it is what programs the beta cells to do better in challenging situations,” said Van de Velde.

Just one injection is not enough; requires long-term use. The altered beta cells produce more insulin and become more resistant to toxic blood sugar levels.
To prove their findings, which is part of a study published in PNAS, the Salk team began working on a comparison to prove Med14’s important role. They made minor mutations in the beta cells of the mice’s pancreas, preventing the chemical modifications that normally occur in response to drugs. The number of cells decreases, weakening the production of insulin.
“We basically turned off the switch so it was always off; it wasn’t responsive to the GLP-1 drug anymore,” Van de Velde said.
Although this was proven in mice, the results probably also apply to humans.
These findings have deepened the understanding of the effects of GLP-1 in the human body, there are more questions that need to be answered.
“There is evidence that this effect disappears when you stop taking the drug, but there is also evidence that it does not disappear completely. People lose weight on the drug, off, and lose weight again, but they do not return to their initial level. There is reason to believe that cells enter a more permanent state after long-term use, which is beneficial for patients,” said Van de Velde.
The understanding of this medicine does not correspond to its widespread use. As they are prescribed more, researchers are discovering new potential benefits: inhibiting inflammation, reducing fatty liver disease and potentially reducing the likelihood of neurodegenerative diseases.
Side effects include nausea and other stomach problems, usually corrected by changing the dose or cutting fatty foods from the diet. Patients should consult with their physician or healthcare provider before starting and while taking this medication.
Fully understanding how these drugs work could lead to new therapies, which is why Van de Velde’s work is so important.
“Understanding the mechanisms by which they work will show us how these drugs do everything they can, which could lead to new therapies,” said Van de Velde. “For us, since we are investigating Med14, we will ask if this works throughout the body, like the part of the brain that controls appetite. That area has receptors for GLP-1 and is known to block your urge to eat or desire – it can also block addictive behaviors like alcohol.”
#weight #loss #miracle #drug #doubled #Salk #Institute #studies #benefits